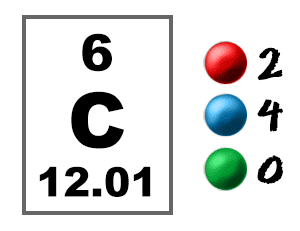 
It has some unique properties that help it to form various substances. Coal tar is a form of carbon itself that is used in making roads.Ĭarbon is an abundant element in nature. Various synthetic compounds such as carbon rubbers, shoe soles, tires of vehicles, etc.Graphite is used in making electrical appliances, pencil leads, etc., and fullerene is used as photosensitizers and drug delivery agents in the body. Like, diamond is used for making tools for cutting or drilling, they are widely used in making jewellery. Allotropes of carbon are used for various purposes.Carbon is present widely in fossil fuels like coal and other organic compounds such as petroleum, natural gas,etc.We release carbon dioxide during respiration and plants inhale it that maintains a complete balance in the form of the carbon cycle in all biological systems and play the most important role in their survival. Carbon dioxide is one of the most important gases present in the atmosphere.They form carbohydrates which provide us energy. Our skin, cells, hair all are made up of carbon. About 18% of the human body is made up of carbon as they behave as building blocks of our body in the form of hydrocarbons.The various uses of carbon are listed below: Carbon is extremely useful in all areas of Science, including chemical, physical, and biological research. To put it another way, we can say that our lives are “carbon-based.” Almost all biological compounds have it as their backbone. Learn Exam Concepts on Embibe Uses of Carbon: Atomic Number of Carbon, Carbon Melting Point Coal is the main source of energy used in thermal power plants. For example, carbon fibres are used to make tennis rackets, fishing rods, and aeronautical devices, carbon black is used for cutting and drilling rocks, and diamonds are used in cutting and drilling rocks. Several different forms of carbon are also used in multiple fields. Read more to learn about the uses of carbon, carbon melting point, and more. Food, plants, timber, fossil fuels, methane gas, and petroleum products all contain it. The human body, for example, contains significant amounts of carbon. These carbon forms are utilised to make a variety of things that we use every day. Because of its tetravalency, carbon possesses a unique self-linking property known as ‘catenation.’ Catenation transforms carbon into a range of organic compounds.Ĭoal, charcoal, and their allotropic form (diamond, graphite, and fullerene) are all examples of carbon in nature. The atomic number of carbon is 6 and belongs to Periodic Table group 14. Uses of Carbon: Uses of carbon by man have had diverse reasons since the dawn of time, and it is a plentiful element in the Earth’s crust.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
